Multicellular migration and environment-stored memory
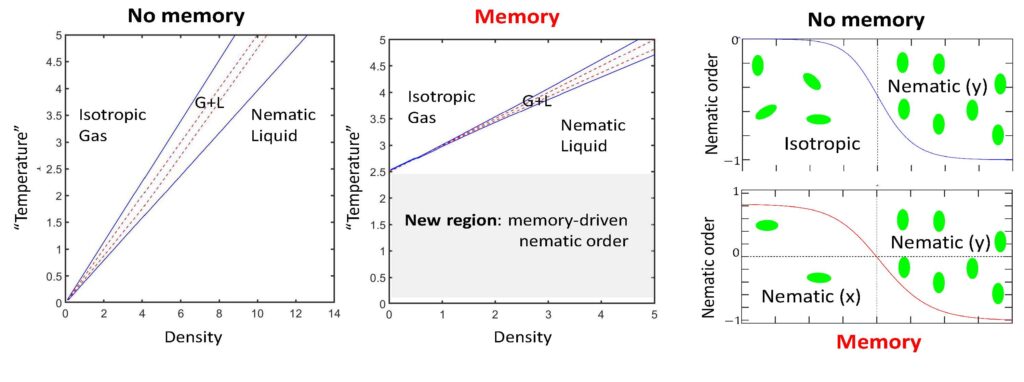
Cell migration is fundamental to biological processes such as development, wound healing, and metastasis. Recent experiments demonstrate that migrating cells induce chemical and mechanical changes in their environment, which act as cues for subsequent migration. These include the secretion of cellular trails and the alignment of collagen fibers in the extracellular matrix. We refer to these phenomena as “environment-stored memory.” From a physics perspective, cell migration is an exemplary case of active dynamics. However, studies often focus on idealized systems, overlooking memory effects and the interconnected dynamics of the environment. Our research aims to elucidate the effects of environment-stored memory on multicellular migration and active-matter dynamics in general.
To this end, we developed a physical framework that treats cells and their environment on equal footing as a multi-component, active system. We demonstrated how cell-environment feedback, both mechanical and chemical, can qualitatively change cellular and tissue organization, as well as multicellular migration. More generally, our studies show that memory qualitatively modify the isotropic-ordered phase diagram in active systems and their characteristic dynamics.
In the long run, our findings can advance the fundamental understanding of active systems and promote the early detection and treatment of metastasis.
Selected publications
- R. M. Adar and J. F. Joanny, “Permeation instabilities in active polar gels,” Phys. Rev. Lett. 127, 10 (2021).
- R. M. Adar and J. F. Joanny, “Environment-stored memory in active nematics and extracellular matrix remodeling,” Phys. Rev. Lett. 133, 118402 (2024).
Cellular volume regulation
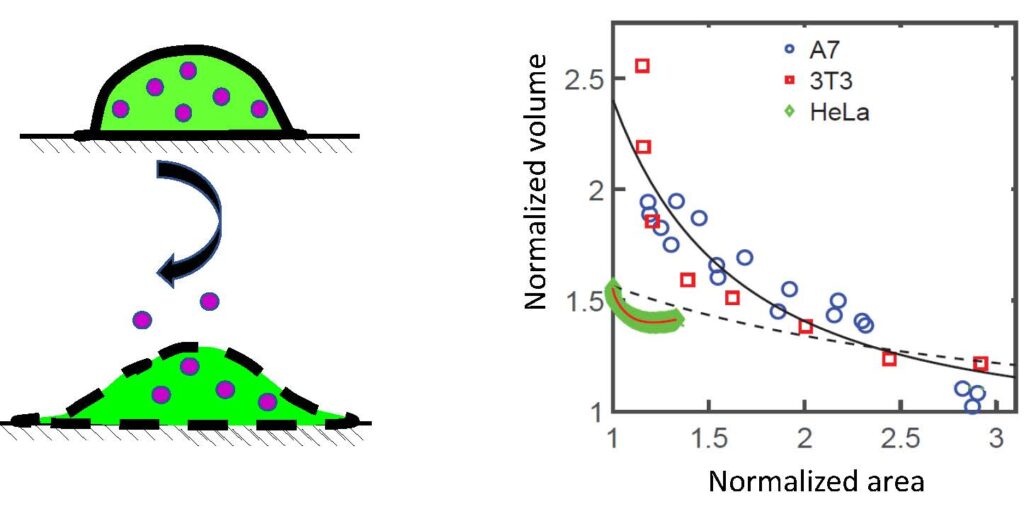
Cellular volume regulation is an integral part of homeostasis and its physical understanding is essential for the understanding of cellular function in health and disease. Cellular volume responds to changes both in the extra-cellular osmolarity and in the properties of underlying substrates. Up to tens of minutes, volume regulation is dominated by water and ion transport through the cell membrane, referred to as “pump-leak”. Ions diffuse passively (“leak”) through membrane-bound ion channels and are transported actively through energy-consuming pumps. The volume dynamics are determined from the water flux, driven by pressure differences across the cell membrane.
We recently developed a theory that explains cellular volume dynamics in terms of an effective ionic chemical potential, set by active pumping. Our theory explains well several recent, independent experiments where cellular volume was shown to decrease during spreading. We found that the effect is due to mechano-sensitive ion transporters that open during spreading, and allow for an ionic flux out of the cell. We are currently applying a similar framework to provide novel predictions of cellular volume regulation in response to two interesting and biologically relevant stimuli: (A) shocks, either osmotic or during immune response to pathogens; (B) stochastic tension changes during 2D migration.
Selected publications
- R. M. Adar and S. A. Safran, “Active volume regulation in adhered cells,” PNAS 117, 5604-5609 (2020).
- R. M. Adar, A. S. Vishen, J. F. Joanny, P. Sens, and S. A. Safran, “Volume regulation in adhered cells: roles of surface tension and cell swelling,” Biophysical J. 122, 506-512 (2023).
Concentrated ionic fluids
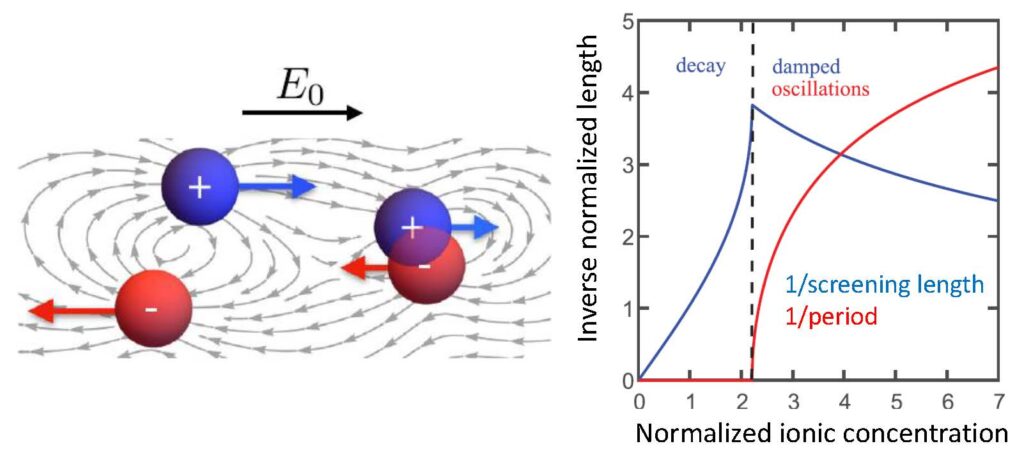
Electrolytes are ubiquitous in nature and industry, and can be found in each cell of our bodies, as well as the batteries of our cellular phones. While the theory of dilute electrolytes is well established, the basic physical properties of concentrated electrolytes and ionic liquids are not well understood. Namely, recent experiments imply that electrostatic interactions are surprisingly stronger in more concentrated electrolytes, while they are expected to be weaker due to screening.
The study of concentrated ionic fluids is challenging due to the combined contributions of Coulombic interactions, entropy, charge correlations, and short-range steric repulsion. In recent works, we have highlighted the role of the latter two in determining basic physical properties of concentrated ionic fluids, including their dielectric constant, screening length, charge oscillations and conductivity. However, these theories do not directly treat strong correlations beyond mean field, which are especially important in ionic liquids. In order to solve this problem, we aim to formulate a complete “strong coupling” theory of ionic fluids, using tools from quantum field theory. Such a theory is in high demand due to the growing industrial applications of these fluids in batteries and super-capacitors.
Selected publications
- Y. Avni, R. M. Adar, D. Andelman, and H. Orland, “Conductivity of concentrated electrolytes,” Phys. Rev. Lett. 128, 098002 (2022).
- R. M. Adar, S. A. Safran, H. Diamant, and D. Andelman, “Screening Length for Finite-size Ions in Concentrated Electrolytes,” Phys. Rev. E 100, 042615 (2019).
